|
11/19/2023 0 Comments 1 bar to psi at sea level
Pascal is the SI unit of pressure and 1 atm=101,325 pascals. The unit of pressure measure of the average atmospheric pressure at sea level is the standard atmosphere, written as atm. The Standard Atmospheric Pressure is based on the average pressure at sea level at 45 degree north latitude.Īt sea level,273 oK(0oc), the Standard Atmospheric Pressure is as follows: temperature and altitude above sea level affect air pressure. Atmospheric PressureĪtmospheric pressure is the pressure in the surrounding air at close to the earth’s surface. When gauge pressures are above atmospheric pressure, they are positive when they are below atmospheric pressure, they are negative. Because practically all pressure gauges show zero when exposed to the atmosphere, the later pressure scale was established. Gauge pressure is when pressure is measured in relation to atmospheric pressure 14.7 gWhen the pressure in a system exceeds the local atmospheric pressure, patm, the phrase gauge pressure is used. 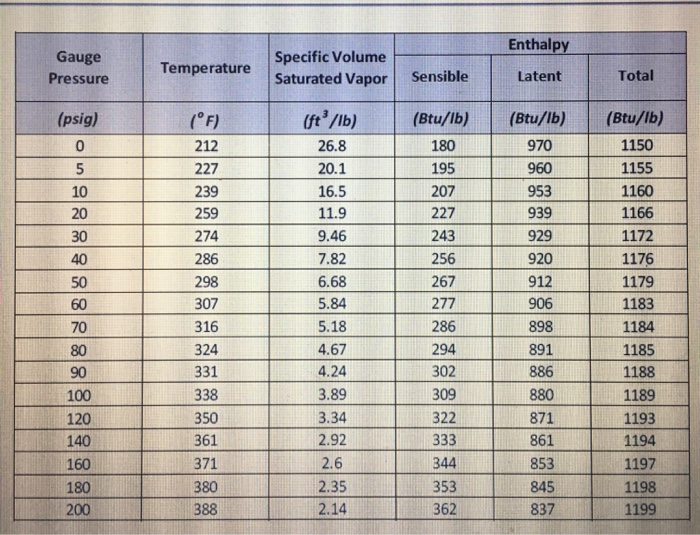
any pressure measuring in air at sea level will be corrected. Because atmospheric pressure at sea level is roughly 101.3 kPa 14.7psi. 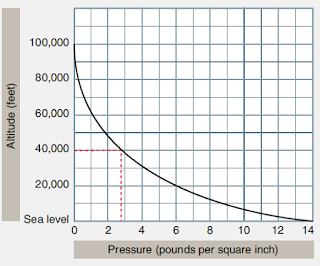
The unit of measurement is pounds per square inch absolute (psia), which indicates that the pressure is measured relative to a vacuum rather than the ambient atmospheric pressure. Types of Pressure Absolute PressureĪbsolute pressure is defined as pressure measured in relation to a perfect vacuum (psia). Atmospheric pressure is typically expressed in millibars (kPa). The standard sea level pressure is defined as 1013 mbar, 1.013 bar, or 101.3 (kPa). One bar equal 100 000 Pa, which is considerably less than the average atmospheric pressure at sea level (1 bar=0.9869 atm). It’s about the same as the air pressure at sea level on Earth. The bar is a pressure unit that equals 100 kilopascals. It only works with liquid and gaseous systems. Pressure is a constant intense variable in a closed system. The forces coming from all of these contacts produce the pressure exerted by a system on its surroundings. As molecules collide with the walls, forces are exerted that attempt to push the barriers outward. In general, pressure or the force exerted per unit area on a substance’s borders, is created by collisions between the substance’s molecules and the system’s boundaries. Pascal is stated as a 1N force applied to a unit area. In the SI system, the Newton per square metre, or pascal, is the standard unit for pressure (Pa). The forces acting per unit area on the edges of a substance is measured as pressure. It’s about the same as the average air pressure at sea level on Earth 1 atmosphere=normal atmospheric pressure. It’s sometimes referred to as a “reference” or “standard” pressure. The standard atmosphere (known as atm) is a pressure unit with a value 101,325 Pa. The bar is extensively used in industry and meteorology, and a barometer is a tool used in atmospheric science to measure atmospheric pressure. 
It is not a unit of measurement in the International System of Units (SI). There are exceptions however, such as water's density increasing between 0☌ and 4☌.īelow is a table of units in which density is commonly expressed, as well as the densities of some common materials.The bar is a metric pressure unit. Increases in temperature tend to decrease density since the volume will generally increase. An increase in pressure decreases volume, and always increases density. However, when regarding gases, density is largely affected by temperature and pressure. In the case of solids and liquids, the change in density is typically low. Note that density is also affected by pressure and temperature. It is useful to carefully write out whatever values are being worked with, including units, and perform dimensional analysis to ensure that the final result has units of mass There are many different ways to express density, and not using or converting into the proper units will result in an incorrect value. However, it is important to pay special attention to the units used for density calculations. The calculation of density is quite straightforward.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
